EMVS/IMVS
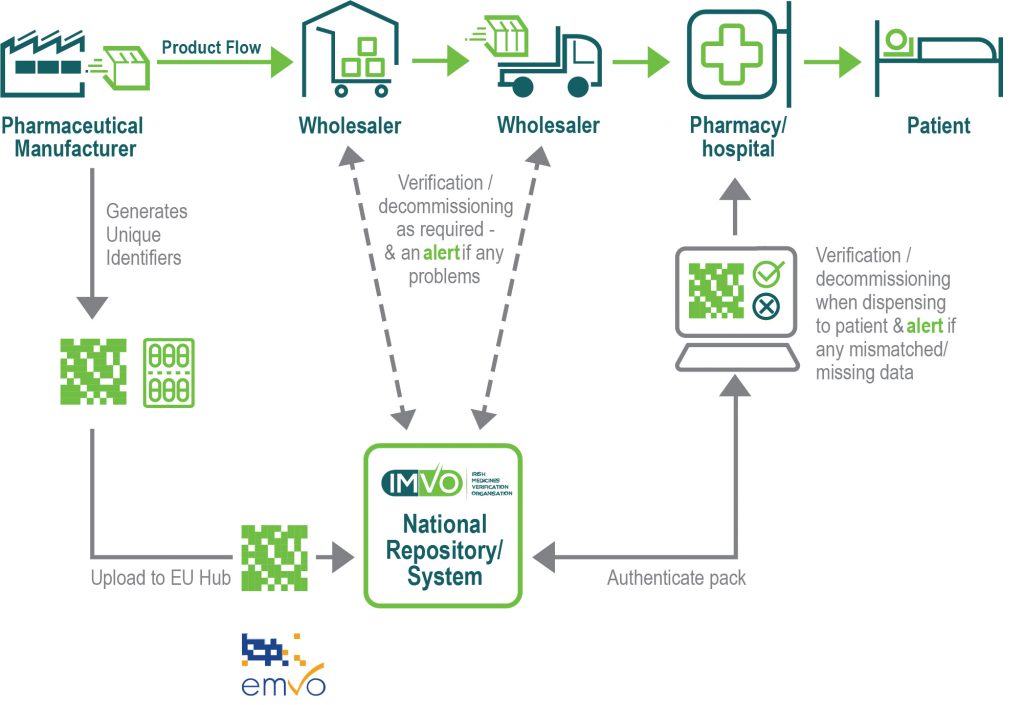
The European Medicines Verification System (EMVS) is a pan-European system covering all EU and EEA countries (except Greece and Italy who will join later). It comprises a central EU Hub managed by the European Medicines Verification Organisation (EMVO) and national medicines verification systems in 30 countries across Europe. IMVO is the national medicines verification organisation for Ireland and operates the Irish Medicines Verification System.
The verification of the unique identifiers of packs in a market takes place in the relevant national system (or repository). The EU Hub connects the national systems in order to make them interoperable. Pharmaceutical manufacturers have a secure connection to the EU Hub to upload unique identifier data to the national systems where the packs will be sold.

 EMVS/IMVS
EMVS/IMVS